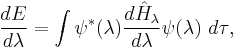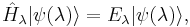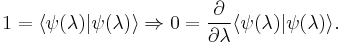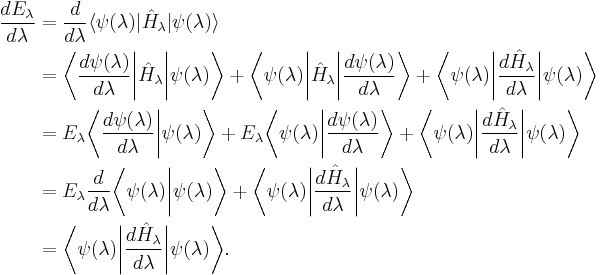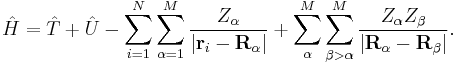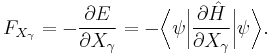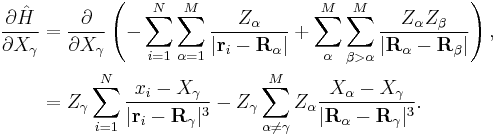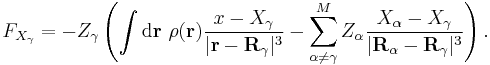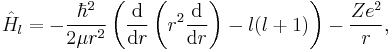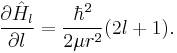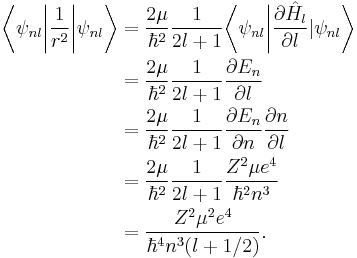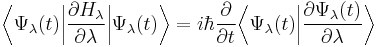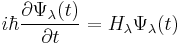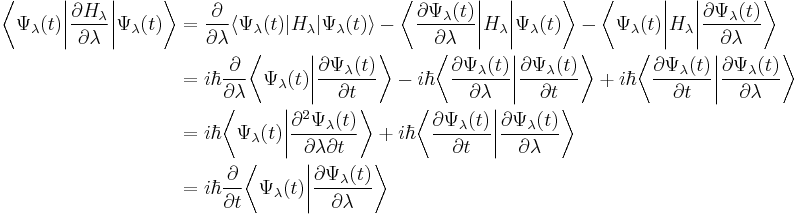Hellmann–Feynman theorem
In quantum mechanics, the Hellmann–Feynman theorem relates the derivative of the total energy with respect to a parameter, to the expectation value of the derivative of the Hamiltonian with respect to that same parameter. Its most common application is in the calculation of forces in molecules (with the parameters being the positions of the nuclei) where it states that once the spatial distribution of the electrons has been determined by solving the Schrödinger equation, all the forces in the system can be calculated using concepts of classical electrostatics.
The theorem has been proven independently by many authors, including Paul Güttinger (1932),[1] Wolfgang Pauli (1933),[2] Hans Hellmann (1937)[3] and Richard Feynman (1939).[4]
The theorem states
where
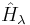 is a Hamiltonian operator depending upon a continuous parameter
is a Hamiltonian operator depending upon a continuous parameter 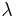 ,
,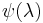 is a wavefunction (eigenfunction) of the Hamiltonian, depending implicitly upon
is a wavefunction (eigenfunction) of the Hamiltonian, depending implicitly upon 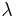 ,
,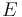 is the energy (eigenvalue) of the wavefunction,
is the energy (eigenvalue) of the wavefunction,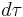 implies an integration over the domain of the wavefunction.
implies an integration over the domain of the wavefunction.
Contents |
Proof
The proof of the Hellmann–Feynman theorem requires that the wavefunction be an eigenfunction of the Hamiltonian under consideration. However, the wavefunction does not need to be exact. For instance in Hartree–Fock theory the wavefunction is a relatively poor approximation to the true wavefunction, but because it is variationally optimized with respect to the Hamiltonian, the Hellmann–Feynman theorem can be applied. Notable example of where the Hellmann–Feynman is not applicable is for example finite-order Møller–Plesset perturbation theory, which is not variational.[5]
The proof also employs an identity of normalized wavefunctions – that derivatives of the overlap of a wavefunction with itself must be zero. Using Dirac's bra-ket notation these two conditions are written as
The proof then follows through an application of the derivative product rule to the expectation value of the Hamiltonian viewed as a function of λ:
For a deep critical view of the proof see [6]
Example applications
Molecular forces
The most common application of the Hellmann–Feynman theorem is to the calculation of intramolecular forces in molecules. This allows for the calculation of equilibrium geometries – the nuclear coordinates where the forces acting upon the nuclei, due to the electrons and other nuclei, vanish. The parameter λ corresponds to the coordinates of the nuclei. For a molecule with 1 ≤ i ≤ N electrons with coordinates {ri}, and 1 ≤ α ≤ M nuclei, each located at a specified point {Rα={Xα,Yα,Zα)} and with nuclear charge Zα, the clamped nucleus Hamiltonian is
The force acting on the x-component of a given nucleus is equal to the negative of the derivative of the total energy with respect to that coordinate. Employing the Hellmann–Feynman theorem this is equal to
Only two components of the Hamiltonian contribute to the required derivative – the electron-nucleus and nucleus-nucleus terms. Differentiating the Hamiltonian yields[7]
Insertion of this in to the Hellmann–Feynman theorem returns the force on the x-component of the given nucleus in terms of the electronic density (ρ(r)) and the atomic coordinates and nuclear charges:
Expectation values
An alternative approach for applying the Hellmann–Feynman theorem is to promote a fixed or discrete parameter which appears in a Hamiltonian to be a continuous variable solely for the mathematical purpose of taking a derivative. Possible parameters are physical constants or discrete quantum numbers. As an example, the radial Schrödinger equation for a hydrogen atom is
which depends upon the discrete azimuthal quantum number l. Promoting l to be a continuous parameter allows for the derivative of the Hamiltonian to be taken:
The Hellmann–Feynman theorem then allows for the determination of the expectation value of 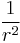 for hydrogen-like atoms:[8]
for hydrogen-like atoms:[8]
Van der Waals forces
In the end of Feynman's paper, he states that, "Van der Waals's forces can also be interpreted as arising from charge distributions with higher concentration between the nuclei. The Schrödinger perturbation theory for two interacting atoms at a separation R, large compared to the radii of the atoms, leads to the result that the charge distribution of each is distorted from central symmetry, a dipole moment of order 1/R7 being induced in each atom. The negative charge distribution of each atom has its center of gravity moved slightly toward the other. It is not the interaction of these dipoles which leads to van der Waals's force, but rather the attraction of each nucleus for the distorted charge distribution of its own electrons that gives the attractive 1/R7 force".
Hellmann–Feynman theorem for time-dependent wavefunctions
For a general time-dependent wavefunction satisfying the time-dependent Schrödinger equation, the Hellmann-Feynman theorem is not valid. However, the following identity holds:
For
Proof
The proof only relies on the Schrödinger equation and the assumption that partial derivatives with respect to λ and t can be interchanged.
Notes
- ^ Güttinger, P. (1932). "Das Verhalten von Atomen im magnetischen Drehfeld". Z. Phys. 73 (3–4): 169. Bibcode 1932ZPhy...73..169G. doi:10.1007/BF01351211.
- ^ Pauli, W. (1933). "Principles of Wave Mechanics". Handbuch der Physik. 24. Berlin: Springer. p. 162.
- ^ Hellmann, H (1937). Einführung in die Quantenchemie. Leipzig: Franz Deuticke. p. 285. OLOL21481721M.
- ^ Feynman, R. P. (1939). "Forces in Molecules". Phys. Rev. 56 (4): 340. Bibcode 1939PhRv...56..340F. doi:10.1103/PhysRev.56.340.
- ^ Jensen, Frank (2007). Introduction to Computational Chemistry. West Sussex: John Wiley & Sons. p. 322. ISBN 0-470-01186-6.
- ^ Carfì, David (2010). "The pointwise Hellmann–Feynman theorem". AAPP Physical, Mathematical, and Natural Sciences 88 (1): no. C1A1001004. doi:10.1478/C1A1001004. ISSN 1825-1242.
- ^ Piela, Lucjan (2006). Ideas of Quantum Chemistry. Amsterdam: Elsevier Science. p. 620. ISBN 0-444-52227-1.
- ^ Fitts, Donald D. (2002). Principles of Quantum Mechanics : as Applied to Chemistry and Chemical Physics. Cambridge: Cambridge University Press. p. 186. ISBN 0-521-65124-7.